
* Autor de contacto:
jmilone@agro.uba.ar
Recibido:
19-09-23
Recibido con revisiones: 09-11-23
Aceptado:
14-11-23

Guadua chacoensis BAMBOO BIOCHAR (POACEAE, BAMBUSEAE)
AFFECTED HORTICULTURAL SPECIES IN A SALINE-ALKALINE SOIL
Julieta Milone 1 3 *, Cecilia Casas 2 3 4, Andrea Susana Vega 1 3
1 Universidad de Buenos Aires, Facultad de Agronomía, Departamento de Recursos Naturales y Ambiente,
Cátedra de Botánica General. Buenos Aires, Argentina.
2 Universidad de Buenos Aires, Facultad de Agronomía, Departamento de Recursos Naturales y Ambiente,
Cátedra de Edafología,. Buenos Aires, Argentina.
3 Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET)
4 Instituto de Investigaciones Fisiológicas y Ecológicas Vinculadas a la Agricultura (IFEVA). Buenos Aires, Argentina.
ABSTRACT
Biochar is being used with increasing frequency as a soil remediator. However, its impact depends on soil limitations, biochar source and concentration applied. We studied Guadua chacoensis bamboo biochar as a saline-alkaline soil amendment. Aqueous solutions with 0, 2.5 and 5% of bamboo biochar (BC) were used to determine their influence on seed germination rate and radicle length in two horticultural species moderately tolerant to salinity and alkalinity: arugula (Eruca sativa) and radish (Raphanus sativus). Also, we amended a saline-alkaline soil with 0, 0.5 and 2.5% of BC to evaluate its effect on plant height, leaf and root biomass of both horticultural crops. The application of 2.5 and 5% BC solution decreased arugula germination in 3.14% and 5.2%, respectively, compared to 0% BC. The addition of BC decreased radicle length in both species and such reduction was more significant with the higher BC concentration. The addition of 0.5% and 2.5% BC as amendment in a saline-alkaline soil decreased arugula height in 27.32% and 50.33%, respectively, compared to 0% BC, resulting in smaller plants with less leaf and root biomass. The BC amendment did not affect leaf biomass, but 2.5% BC reduced root biomass in 53.85% compared to 0% BC. These results seem to be influenced not only by the BC inherent characteristics but also by the increase in soil pH, caused by the addition of biochar. Further studies may evaluate the impact of BC addition with different properties on saline-alkaline soils, and its use as an amendment in the remediation of acid soils.
Keywords: soil amendment; seed germination; plant growth.
EL BIOCARBÓN DEL BAMBÚ guadua chacoensis (POACEAE, BAMBUSEAE)
AFECTÓ A ESPECIES HORTÍCOLAS EN SUELO SALINO-SÓDICO
RESUMEN
El biocarbón se utiliza como remediador de suelos cada vez con más frecuencia. Sin embargo, el impacto en el suelo depende de las limitaciones edáficas, el material empleado para obtener el biocarbón y la concentración aplicada. Se estudiaron los efectos del biocarbón de bambú de Guadua chacoensis sobre la germinación, y como una técnica de enmienda de suelos salino-sódicos. Se utilizaron soluciones acuosas con 0, 2,5 y 5% de biocarbón de bambú (BC) para determinar el porcentaje de germinación de semillas y la longitud de radícula en dos especies hortícolas: rúcula (Eruca sativa) y rabanito (Raphanus sativus). Además, se incorporó 0, 0,5 y 2,5% de BC a un suelo salino-sódico para evaluar el efecto sobre ambas especies, las cuales son moderadamente tolerantes a la salinidad. En cada especie se evaluó la altura de la planta, la biomasa foliar y radicular. Las soluciones de 2,5 y 5% BC disminuyeron la germinación de la rúcula en un 3,14% y 5,2%, respectivamente, en comparación con el control. La adición de BC disminuyó la longitud de la radícula en ambas especies, siendo la reducción más significativa con la mayor concentración de BC. La adición de 0,5% y 2,5% de BC disminuyó la altura de la rúcula en un 27,32% y 50,33%, respectivamente, en comparación con el control, lo que resultó en plantas más pequeñas con menos biomasa de hojas y raíces. La adición de biocarbón no afectó la biomasa foliar, pero en el tratamiento 2,5% BC se redujo la biomasa de raíces en un 53,85% en comparación con el 0% BC. Además de las características inherentes del biocarbón, estos resultados estarían influenciados por el aumento del pH del suelo, provocado por la adición del BC. Se propone evaluar el biocarbón con distintas propiedades en suelos salino-sódicos, y como enmienda en la remediación de suelos ácidos.
Palabras clave: enmienda de suelos; germinación de semillas; crecimiento de las plantas
INTRODUCTION
Salinity and alkalinity due to natural causes or unsustainable anthropogenic practices (Guida-Johnson et al., 2017; Madrigal, 2016) are considered some of the most important soil problems in the world (Aimetta et al., 2020). Halomorphic soils present limitations for agricultural activities, reducing crop yields (Sanchez et al., 2015). Nearly 424 Mha of worldwide superficial soils (0-30 cm) are affected by some level of salinity and alkalinity (Food and Agriculture Organization of the United Nations [FAO], 2021). Salinity impacts plant growth by reducing water uptake due to osmotic imbalances in the root zone (Alcivar et al., 2018; Hussain et al., 2017) and increasing aluminum toxicity (Dai et al., 2017). Alkaline soils accumulate excessive exchangeable Na+ in soil solution, negatively affecting soil structure by clay dispersion and compaction (Hasana et al., 2022) and restricting the movement of water and air through the soil profile. It also alters pH and nutrient availability with a decrease in crop yields (Yu et al., 2010).
Biochar is a solid material obtained by the thermochemical conversion of biomass in an oxygen-limited environment (International Biochar Initiative [IBI], 2012) with potential benefits, such as carbon sequestration and environmental management (i.e., soil improvement, waste treatment, climate change mitigation, and energy production; Lehmann & Joseph, 2015). In the last few years, an increasing focus on biochar has been reported for soil remediation (Nath et al., 2022). Based on their wide availability, different agricultural and forest valueless wastes [i.e., corn straw (Naeem et al., 2016), shell peanut (Sathe et al., 2021), forest branches (Ke et al., 2018), among others] are used for biochar production. Ahmad et al. (2014) reported that the addition of different biochar to soils increased seed germination, plant growth, and crop yields. Particularly, the addition of 1.5% (w/w) of corn cobs biochar increased maize germination in 13% in a sandy loam soil (Ali et al., 2021). Moreover, biochar derived from pelletized poultry manure improved lettuce growth on a nutrient deficient soil (Gunes et al., 2014) and promoted rice crop by reducing Al3+ uptake by roots in acid soils (Shetty & Prakash, 2020). Several studies have reported soil improvements after the addition of biochar, such as reducing soil bulk density (Muñoz et al., 2016) and increasing water retention (Glaser et al., 2002) due to its organic carbon content (60-80%) and highly active surface area (Yu et al., 2019). For these reasons, biochar arises as an environmentally friendly amendment to improve soil quality and productivity (Sathe et al., 2021).
Biochar application was assessed in various soil types (Abdullaeva, 2014), but few studies have been carried out under saline or alkaline conditions (Zhao et al., 2020). Biochar application reduced salt stress and improved plant growth in relation to Na+ removal and adsorption onto biochar surfaces (Yu et al., 2019). However, its effectiveness depends on soil physicochemical properties, pyrolysis conditions (temperature, aeration, and time), the amount of biochar applied, the specific surface area, pore size distribution, ion exchange capacity, and the taxonomic identity of the species used to produce the biochar (Ahmad et al., 2014; Alchouron et al., 2020).
Bamboo culms are suitable for biochar production (Wang et al., 2012; Zhang, 2009) and bamboo BC has proven to be a good adsorbent for air, water, and soil purification (Wang et al., 2012). In addition, bamboo is considered a sustainable alternative for biochar production given its rapid regeneration (Orosco Gutierrez & De Lira Fuentes, 2020). In particular, Guadua chacoensis (Rojas Acosta) Londoño & P. M. Peterson is a native woody bamboo from Bolivia, Paraguay, Uruguay, Brazil, and Argentina (Lizarazu & Vega, 2012; Vega & Rúgolo, 2016). This bamboo grows vegetatively for 28 years, flowers, and dies, successfully reproducing through its caryopses (Vega & Cámara Hernández, 2008). Interestingly, recent studies showed that G. chacoensis Fe3O4 nanoparticle-covered biochar remediated arsenic contaminated water, reducing arsenic almost completely and reaching concentration levels below those recommended by the World Health Organization (WHO; Alchouron et al., 2020, 2021). Although bamboo has long been cultivated and used in Southeastern Asia, its cultivation in tropical and subtropical America is only recent (Londoño, 2009), especially in the case of G. chacoensis. For this reason, the effects of G. chacoensis biochar as a soil amendment have not been explored yet.
In order to study the effects of G. chacoensis biochar as a remediation technique for saline-alkaline soils, we investigated its impact on the germination and performance of two horticultural species: radish (Raphanus sativus L.) and arugula [Eruca vesicaria (L.) Cav. subsp. sativa (Mill.) Thell.] which are moderately tolerant to salinity and alkalinity. We hypothesize that (i) biochar contributes with naturally adsorbed essential elements that can be released into the biochar solution, improving seed germination rate; (ii) the biochar surface adsorbs and removes Na+ in soil, thus improving soil structure and plant performance. To test these hypotheses, we evaluated G. chacoensis biochar at increasing concentrations through two experiments: the first one focused on germination assessment in vitro, while the second one explored plant establishment and biomass production of both species when growing in a saline-alkaline soil.
MATERIALS AND METHODS
Plant species
Two horticultural species belonging to the Brassicaceae family were selected (i.e., Raphanus sativus and Eruca vesicaria subsp. sativa), since they are annual crops with life cycles that are completed in 20-60 days (Sangiacomo et al., 2002a, b). Both species are moderately tolerant to salinity and alkalinity (Foti & Lallana, 2005; Rahman et al., 2017; Sangiacomo et al., 2002a, b). Seeds were obtained through the Instituto Nacional de Tecnología Agropecuaria (INTA Castelar, Buenos Aires, Argentina).
Bamboo Biochar (BC)
Discarded culms (i.e., without commercial value) of the native bamboo G. chacoensis (Poaceae, Bambusoideae, Bambuseae) were used as a raw material for biochar production. Samples were obtained from three different geographic regions in Argentina: Buenos Aires, Corrientes, and Misiones. Voucher specimens are deposited at Gaspar Xuarez Herbarium (BAA- code after Thiers 2023), from the following locations in Argentina: Buenos Aires: Capital Federal, Cult. Jardín Botánico de la Facultad de Agronomía, A. S. Vega 17 (BAA); Corrientes: Dpto. Capital, route 12, 3 km from Riachuelo, 27º34′S, 58º44′W, 60 m, A. S. Vega 23 (BAA); Misiones, Dpto. Guaraní, El Soberbio, 27°16′20″S, 54°11′36″O, A. S. Vega 104 (BAA). The material was dried at room temperature and ground to a 0.5-1 cm particle size using chipping machines (Troy Bilt, CS 4265, Canada and EcoGreenChip, K3, Argentina). Then, the homogenised milled material was subjected to slow pyrolysis carbonization at 700 ºC for 2 h in a tubular muffle furnace (O.R.L., Argentina) inside a steel reactor (AISI 310) under a continuous nitrogen flow (10 mL/min) according to Alchouron et al. (2020). The resulting BC was ground, sieved to particle size range of 150–300 μm, and stored in hermetic plastic bags.
Soil material origin and preparation
The saline-alkaline soil material (i.e., with excessive soluble salts and exchangeable sodium) used in this study came from topsoil (0–10 cm) of a horticultural field (34° 58’ 43.023”, S 58° 2’ 38.132” W; La Plata, Buenos Aires). The soil material was air-dried, ground to pass through a 2 mm sieve, and homogenised. The soil material was characterised according to electrical conductivity (EC) and pH (in 1:2.5 soil and distilled water), cation exchange capacity (CEC; Sumner and Miller, 1996), sodium concentration in the extract of saturation and texture (ESP; Bouyoucos, 1951), oxidizable organic carbon (OC; IRAM/SAGyP, 2016), and organic matter (OM; Walkley & Black, 1934). Then, the soil material was mixed with different biochar proportions: 0, 0.5 or 2.5% to generate three treatments, according to previous studies (Ali et al. 2021; Hussien Ibrahim et al. 2020)
Seed germination
Two in vitro assays were conducted to evaluate biochar effect on germination of radish and arugula seeds, isolating the effect of soil conditions. For this purpose, Petri dishes were maintained into a germination chamber under controlled temperature, light, and humidity conditions. Each Petri dish contained 10 seeds and 3 mL of an aqueous solution of distilled water + biochar in different concentrations according to the following treatments: 0, 2.5, and 5% BC with 10 replicates for each treatment and species, using a completely randomised design. Seed germination percentage (%) and radicle length (in cm) were daily monitored for 3 days.
Plant species establishment and growth
Two experiments under controlled conditions and completely randomised design were performed in a greenhouse trial at the Instituto de Investigaciones Fisiológicas y Ecológicas Vinculadas a la Agricultura (IFEVA, CONICET). The purpose of these experiments was to evaluate the effect of biochar application in a saline-alkaline soil material on the capacity of seeds to germinate. Plastic pots (1 L) were filled with soil and biochar mixed in different concentrations according to the following treatments: 0, 0.5 and 2.5% BC, and four seedlings of each species were transplanted to individual pots (8 replicates and 24 pots for each vegetable species). In order to homogenise plant size along the experiment, only two seedlings per pot were maintained and harvested 30 days after sowing. The number of replicates for the arugula growth experiment was unbalanced due to the lack of germination in some pots, so no plant was considered for the plant growth test. Pot positions in the greenhouse were daily and fully randomized. Growth indicators included plant height (cm.plant-1), leaf biomass (g.plant-1), and root biomass (g.pot-1). Plant height was measured from the base of the stem at the soil surface to their apex. Plants were cut at the soil surface, and everything below this point was considered as roots, which were thoroughly washed to remove adhering soil particles. Root and shoot biomass were dried at 60 °C for 24 h prior to weighing for dry biomass.
Soil determinations
Once the experiment was completed, soil samples from each pot were characterized to analyze the effect of bamboo biochar on soil. At the end of the experiments, the pH and electrical conductivity (EC) were measured in all dry and sieved soil samples (2 mm) in a 1:2.5 solution in distilled water (Altronix Mod EZDO-PC pH meter and Lutron CD-4318SD conductivity meter).
Data analysis
Seed germination was analysed through a mixed-effects model with binomial distribution of errors, Bernoulli distribution (glmer in lme4 in R; Bates et al., 2015), while the length of the radicle was analysed through a mixed-effects model with normal distribution of errors (lme of the nlme package in R; Pinheiro et al., 2023). In both cases, the random effect considered the seeds nested in the Petri dishes. Biochar concentration, time and its interaction were considered as fixed effects. For normal error distribution models, temporal autocorrelation was assessed with an ACF plot. When necessary, the first order autocorrelation function (corAR1) was included in the models that evaluated radicle length. The establishment of the seedlings was analysed through a mixed-effects model with binomial distribution of errors, Bernoulli distribution (glmer in lme4 in R; Bates et al., 2015), considering the nesting of the plants in pots.
Growth indicators (i.e., plant height, leaf biomass, and root biomass) and soil parameters (i.e., pH and EC) were analyzed through fixed effect generalized linear models squares (gls) with a normal distribution of errors. Both plant height and dry biomass of leaves and roots were analysed with mixed-effects models that considered nesting of plants in pots (lme of nlme package in R; Pinheiro et al., 2023). In all normal distribution models, the homogeneity of the variances was evaluated by residual plots as a function of adjusted values and treatments. When necessary, variance functions for groups (verIdent) or power (varPower) were applied. The normal distribution of the residuals was evaluated using quantile theoretical distribution plots (Q-Q plot). Models with a binomial distribution were evaluated using the dispersion parameter phi. All analyses and figures were made in the R-cran environment, version 3.6.1 (R Development Core Team, 2019), and with RStudio; version 1.2.1335 (RStudio, 2019).
RESULTS
Soil characteristics
The soil material was a silt loam, with a high percentage of exchangeable sodium, and adequate organic matter and organic carbon (Table 1). It is considered a moderate saline and alkaline soil following the classification proposed by Sonmez et al. (2018). Pyrolysis at 700 ºC resulted in a highly basic biochar whose elemental composition is presented in Table 1.
Table 1: Soil and G. chacoensis biochar characteristics prior to the experiment. The columns show soil textural class, pH, oxidizable organic carbon (OC), electrical conductivity (EC), cation exchange capacity (CEC), sodium concentration (ESP), organic matter (OM), biochar pH and elemental composition including carbon (C), oxygen (O), hydrogen (H) and sulfur (S).
Tabla 1: Características del suelo y del biocarbón de G. chacoensis previo al inicio de los experimentos. Las columnas muestran la textura del suelo, pH, carbono orgánico oxidable (OC), conductividad eléctrica (EC), capacidad de intercambio catiónico (CEC), concentración de sodio (ESP), materia orgánica (OM), pH y composición elemental del biocarbón, incluyendo carbono (C), oxígeno (O), hidrógeno (H) y azufre (S).
|
Value |
|
|
Soil |
|
|
Textural class |
Silt loam |
|
pH |
8.39 |
|
OC (%) |
2.17 |
|
EC (dS/m) |
1.51 |
|
CEC (cmol/Kg) |
30.91 |
|
ESP (%) |
16.00 |
|
OM (%) |
3.96 |
|
Biochar |
|
|
pH |
9.53 |
|
C (%) |
56.56 |
|
O (%) |
43.00 |
|
H (%) |
0.35 |
|
S (%) |
0.09 |
Arugula and radish germination
The proportion of germinated seeds increased over time in both species (arugula: 𝝌2 = 12.69; p-value < 0.001; radish: 𝝌2 = 7.40; p-value < 0.05; Figure 1 A-B). Nevertheless, BC aqueous solutions reduced germination of arugula (𝝌2 = 6.17; p-value < 0.05) and radish seeds (𝝌2 = 3.61; p-value = 0.16). After three days from the beginning of the experiment, germination of arugula seeds was 3.14% (2.5% BC) and 5.21% (5% BC) lower, compared to the control (0% BC). The addition of biochar also decreased the radicle length of germinated seeds in both species, being arugula (𝝌2 = 288.08; p-value < 0.001; Figure 1 C) more sensitive than radish (𝝌2= 19.42; p-value < 0.001; Figure 1 D). In arugula, radicle length was reduced by 68.50% (2.5% BC) and 80.90% (5% BC), compared to 0% BC (Figure 1 C). In relation to radish, radicle length was reduced by 24.70% (2.5% BC) and 54.50% (5% BC), compared to the 0% BC solution (Figure 1 D).
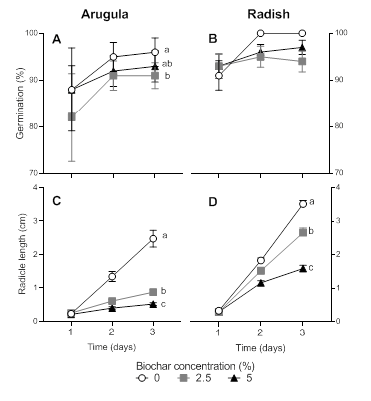
Figure 1: Cumulative percentage of germination (%) and radicle length (cm) in arugula (A, C) and radish seeds (B, D) as a function of time (days). Symbols show average values and bars correspond to the standard error. Different letters show statistically significant differences (p < 0.05) between treatments.
Figura 1: Porcentaje acumulado de germinación (%) y largo de radícula (cm) en semillas de rúcula (A, C) y de rabanito (B, D) en función del tiempo (días). Los símbolos muestran el promedio y las barras el error estándar. Letras distintas indican diferencias estadísticamente significativas (p < 0.05) entre tratamientos.
Arugula and radish growth
The addition of biochar amendment to saline-alkaline soils reduced arugula height (𝝌2 = 9.72; p-value < 0.05; Figure 2 A), leaf biomass (𝝌2 = 8.47; p-value < 0.05; Figure 2 B), and root biomass (𝝌2 = 15.45; p-value < 0.05; Figure 2 C). Plants growing in the saline-alkaline soil amended with 2.5% BC had a reduction of 27.32% in height compared to the control (0% BC). At higher biochar concentration (5% BC) plants reached ca. one-half the height of control plants (50.33%). The addition of 2.5 or 5% BC to the saline-alkaline soil did not alter the height of radish plants (𝝌2 = 2.60; p-value = 0.26; Figure 2 D) and tended to reduce leaf biomass (𝝌2 = 0.56; p-value = 0.76; Figure 2 E). The addition of 0.5% BC to the saline-alkaline soil decreased radish root biomass by 53.85%, with respect to the control (𝝌2 = 7.67; p-value < 0.05; Figure 2 F).

Figure 2: Arugula and radish height (cm.plant-1; A, D), leaf biomass (g.plant-1; B, E), and root biomass (g.pot-1; C, F), respectively. Bars: white: 0% BC, grey: 0.5% BC, black: 2.5% BC. Bars show mean values and the standard error. Different letters indicate statistically significant differences (p < 0.05) between treatments. 0% BC: A-C, E: n= 4; B-D: n=16; F: n=8; 0.5% BC: A-C: n=10; E: n=5; B-D: n=14; F: n=8; 5% BC: A-C: n=10; E: n=6; B-D; n=16; F: n=8.
Figura 2: Altura (cm.planta-1; A, D), biomasa de hojas (g.planta-1; B, E), y biomasa radical (g.macetat-1; C, F), respectivamente, de rúcula y rabanito. Barras: color blanco: 0% BC, gris: 0,5% BC, negro: 2,5% BC. Las barras muestran el promedio y el error estándar. Letras distintas indican diferencias estadísticamente significativas (p < 0.05) entre tratamientos. 0% BC: A-C, E: n= 4; B-D: n=16; F: n=8; 0.5% BC: A-C: n=10; E: n=5; B-D: n=14; F: n=8; 5% BC: A-C: n=10; E: n=6; B-D; n=16; F: n=8.
Soil determination
Soil pH increased slightly but significantly after biochar application in both arugula (𝝌2 = 13.24; p-value < 0.001; Figure 3 A) and radish assays (𝝌2= 59.68; p-value < 0.001; Figure 3 B). The addition of 2.5% BC increased pH in arugula (1%) and radish tests (2%), compared to the control. On the other hand, biochar application did not modify EC in arugula assay (𝝌2 = 0.65; p-value = 0.72; Figure 3 C), but it increased in 7.58% in radish assay when 2.5% BC was applied (𝝌2= 7.97; p-value < 0.05; Figure 3 D).
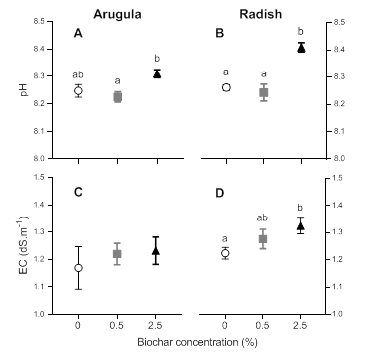
Figure 3: Variation of soil pH and EC (dS.m-1) in arugula (A, C) and radish (B, D) as a function of biochar concentrations. Symbols show average values and bars show the standard error. Different letters express statistically significant differences (p <0.05) between treatments. A, C: BC 0% n=4; BC 0.5%: n=6; BC 2.5%: n=7. B, D: all treatments n=8.
Figura 3: Variación en el pH del suelo y CE (dS.m-1) en rúcula (A, C) y rabanito (B, D) en función de la concentración de biocarbón. Los símbolos muestran el promedio y las barras el error estándar. Letras distintas indican diferencias estadísticamente significativas (p < 0.05) entre tratamientos. A, C: 0% BC n=4; 0.5% BC: n=6; 2.5% BC: n=7. B, D: todos los tratamentos n=8.
DISCUSSION
The use of biochar as a soil amendment has been widely reported (Ahmad et al., 2014; Naeem et al., 2016; Nath et al., 2022; Sathe et al., 2021; among others). In this sense, we used raw material of the native woody bamboo G. chacoensis pyrolyzed at 700 ºC to obtain biochar and test it as a potential saline-alkaline soil amendment. Our results show that biochar addition only affected the germination of arugula seeds, and radicle length decreased in both horticultural species. These effects could be related to soluble phytotoxic substances, such as polycyclic aromatic hydrocarbons, during the production of biochar at temperatures above 700 ºC (Liao et al., 2014; Rogovska et al., 2012;). In this sense, Zhang et al. (2020) reported inhibition of seed germination when using lignin-rich feedstock to obtain biochar nanoparticles at high temperatures (700 °C). To the best of our knowledge, no studies have been conducted to evaluate the capacity of biochars as saline-alkaline soil amendment on arugula plants, which merits further research. Moreover, a complete chemical characterization of these compounds and additional studies are important to elucidate possible toxicity mechanisms.
Not all species are adversely affected by biochar application. Our results showed no significant differences in radish seed germination in the presence of G. chacoensis biochar aqueous solutions, being less sensitive to their effects than arugula. Also, no effects were detected in height and aerial biomass during growth period, in line with the results obtained with biosolid biochar application (Free et al., 2010) or with biochar from forest branches (Ke et al., 2018). Biochar addition produced both positive and negative effects on seed germination and biomass production, depending on feedstock and pyrolysis conditions (e.g., temperature; Awad et al., 2017). These crucial factors influence the physical and chemical properties of biochar, such as pH, specific surface area, pore size distribution, and ion exchange capacity (Ahmad et al., 2014; Sun et al., 2014;). For this reason, the purpose of biochar application should consider final characteristics resulting from the type of raw material and its obtention method (Yu et al., 2019).
On the other hand, radish root biomass and radicle length in the germination test only decreased with the intermediate (0.5%) biochar concentration. A long-term study is required (i.e.: > 2 years) to elucidate the reactions and interactions with soil particles which occur at low speeds (Blanco-Canqui, 2017). Further, combining biochar with organic manure (Yang & Zhang 2022) or inorganic fertilisation (An et al., 2022) could significantly improve radish growth and plant nutrient content, especially when added simultaneously. Mete et al. (2015) reported that biochar addition and NPK fertilization decreased pH and increased P availability, improving soybean biomass production in alkaline soils. Arugula was clearly more sensitive than radish in the presence of biochar, salts, and soil alkalinity, as evidenced by a decrease in all growth indicators (i.e., plant height, leaf, and root biomass), compared to the control. Different possible mechanisms can be associated, such as osmotic effects, ionic toxicity, and nutrient availability (Munns & Tester, 2008) related to soil pH, which is an important factor for plant growth (Maraseni, 2010).
Guadua chacoensis biochar resulted in a highly basic material, and its application increased soil pH. Other studies revealed an increase in soil pH with biochar amendment (Adekiya et al., 2019; Castellón Romero & Andrade Foronda, 2020). In this condition, nutrients such as Cu, Mn, Zn, and Fe are not available for root uptake (Hsieh & Waters, 2016), a possible explanation for the decline in arugula growth. These results are in agreement with Salem et al. (2019), who reported that basic biochar reduced some nutrients availability and plant yield. Biochars obtained at temperatures above 400°C usually have a higher pH and EC than those obtained at lower temperatures, even if the raw material is the same (Singh et al., 2017). Acid functional groups present in the biomass are lost during pyrolysis, resulting in a higher basicity of the biochar (Weber & Quicker, 2018). At high pyrolysis temperature there is an increase in biochar elements, but a decrease in their bioavailability, based on the formation of aromatic structures that contain these elements (Naeem et al., 2016). For this reason, the pyrolysis of G. chacoensis raw material at lower temperatures could improve pH and plant nutrients availability in saline-alkaline soils. Moreover, applying this biochar to acid soils can maximize its benefits (Maraseni, 2010). Soil acidity alleviation is based on biochar buffering capacity and Si effect (Yu et al., 2019). In this sense, G. chacoensis biochar produced at 700ºC has 33.5% of Si (Martínez de Zorzi, 2019), being considered as a Sichar (i.e., biochar that contains more than 5 g of Si per kg; Wang et al., 2019). However, the response of this biochar and the mechanism of action of silica in acid soils is still unknown.
CONCLUSIONS
The present study provides preliminary information about the application of G. chacoensis biochar on a saline-alkaline soil. Two experiments were designed to investigate the response of biochar application under controlled laboratory and greenhouse conditions. The addition of biochar decreased arugula germination and radicle length in both species. No significant differences in radish seed germination, height, and aerial biomass were found. Species like radish may be less sensitive to the effects of biochar. Guadua chacoensis biochar obtained at 700 ºC had no influence on edaphic conditions, germination or biomass production, but increased soil pH. It is proposed that this biochar could be tested in acid soil remediation due to its high pH. Future studies of varying pyrolysis temperatures (probably resulting in biochar with lower pH among other characteristics) are needed to determine if these results are associated with soluble phytotoxic substances or biochar basicity. Also, the combination with other compounds could work as a good amendment technology in saline-alkaline soils.
ACKNOWLEDGMENT
This work was supported by the University of Buenos Aires [grants UBACyT 20020190100206BA and PIDAE 63]. The authors are grateful to Marcela Harris for her valuable help.
REFERENCES
Abdullaeva, Y. (2014). Biochar effects on fertility of saline and alkaline soils (Navoiy region, Uzbekistan). United Nations University Land Restoration Training Programme [final project]. http://www.unulrt.is/static/fellows/document/abdullaeva2014.pdf.
Adekiya, A. O., Agbede, T. M., Aboyeji, C. M., Dunsin, O. & Simeon, V. T. (2019). Effects of biochar and poultry manure on soil characteristics and the yield of radish. Scientia Horticulturae. (243):457–463. doi: 10.1016/j.scienta.2018.08.048
Ahmad, M., Rajapaksha, A. U., Lim, J. E., Zhang, M., Bolan, N., Mohan, D., Vithanage, M., Lee, S. S. & Ok, Y. S. (2014). Biochar as a sorbent for contaminant management in soil and water. A review. Chemosphere 99:19-23. doi: 10.1016/j.chemosphere.2013.10.071
Aimetta, M. B., Muñoz, S. A., Bustos, D., Davidenco, V., Cazorla, C. R., Galarza, C. M. y Salvagiotti, F. (2020). Cuantificación del uso agrícola de suelos con limitantes salinas y sódicas mediante el índice NDVI. Ciencia del Suelo. 38(1):174–186.
Alchouron, J., Navarathna, C., Chludil, H. D., Dewage, N. B., Perez, F., Hassan, E. B., Pittman, C. U. Jr, Vega, A. S. & Mlsna, T. E. (2020). Assessing South American Guadua chacoensis bamboo biochar and Fe3O4 nanoparticle dispersed analogues for aqueous arsenic (V) remediation. Science of Total Environment. 706:135943. doi: 10.1016/j.scitotenv.2019.135943
Alchouron, J., Navarathna, C., Rodrigo, P. M., Snyder, A., Chludil, H. D., Vega, A. S., Bosi, G., Perez, F., Mohan, D., Pittman, C. U. Jr & Mlsna, T. E. (2021). Household arsenic contaminated water treatment employing iron oxide/bamboo biochar composite: an approach to technology transfer. Journal of Colloid Interface Science. 587:767–779. doi: 10.1016/j.jcis.2020.11.036
Alcivar, M., Zurita–Silva, A., Sandoval, M., Muñoz, C. & Schoebitz, M. (2018). Reclamation of saline–sodic soils with combined amendments: impact on quinoa performance and biological soil quality. Sustainability 10(9):3083. doi: 10.3390/su10093083
Ali, L., Xiukang, W., Naveed, M., Ashraf, S., Nadeem, S. M., Haider, F. U. & Mustafa, A. (2021). Impact of biochar application on germination behavior and early growth of maize seedlings: insights from a growth room experiment. Applied Sciences. 11:11666. doi: 10.3390/app112411666
An, N., Zhang, L., Liu, Y., Shen, S., Li, N., Wu, Z., Yang, J., Han, W. & Han, X. (2022). Biochar application with reduced chemical fertilizers improves soil pore structure and rice productivity. Chemosphere 298:134304. doi: 10.1016/j.chemosphere.2022.134304
Awad, Y. M., Vithanage, M., Niazi, N. K., Rizwan, M., Rinklebe, J., Yang, J. E., Ok, Y. S. & Lee, S. S. (2017). Potential toxicity of trace elements and nanomaterials to Chinese cabbage in arsenic– and lead–contaminated soil amended with biochars. Environmental Geochemistry Health 41(5):1777–1791. doi: 10.1007/s10653–017–9989–3
Bates, D., Maechler, M., Bolker, B. & Walker, S. (2015). Fitting linear mixed–effects models using lme4. J. Stat. Softw. 67(1):1–48. doi:10.18637/jss.v067.i01
Blanco–Canqui, H. (2017). Biochar and soil physical properties. Journal of the American Oil Chemists’ Society 81:687–711. doi: 10.2136/sssaj2017.01.0017
Bouyoucos, G. J. (1951). A recalibration of the hydrometer method for making mechanical analysis of soil. Agronomy Journal 43:434–438.
Castellón Romero, D. y Andrade Foronda, D. (2020). Enmiendas orgánicas para la remediación de suelos salino–sódicos del Valle Alto de Cochabamba. Revista de Agricultura (Bolivia) 62:57–64.
Dai, Z., Zhang, X., Tang, C., Muhammad, N., Wua, J., Brookes, P. C. & Xu, J. (2017). Potential role of biochars in decreasing soil acidification – A critical review. Science of Total Environment. 581–582:601–611. doi: 10.1016/j.scitotenv.2016.12.169
Food and Agriculture Organization of the United Nations (FAO). (2021). Global map of salt–affected soils: 1–20. https://www.fao.org/3/cb7247en/cb7247en.pdf
Foti, M. N. y Lallana, V. H. (2005). Bioensayo de germinación con semillas de Eruca sativa Mill. para la detección de salinidad y presencia de herbicida en agua. Revista FABICIB 9:9–16. Spanish.
Free, H. F., McGill, C. R., Rowarth, J. S. & Hedley, M. J. (2010). The effect of biochars on maize (Zea mays) germination. New Zealand Journal of Agricultural Research 53(1):1–4. doi:10.1080/00288231003606039
Glaser, B., Lehmann, J. & Zech, W. (2002). Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal – a review. Biology and Fertility of Soils 35:219–230. doi: 10.1007/s00374–002–0466–4
Guida–Johnson, B., Abraham, E. M. y Cony, M. A. (2017). Salinización del suelo en tierras secas irrigadas: perspectivas de restauración en Cuyo, Revista de la Facultad de Ciencias Agrarias UNCUYO 49(1):205–215.
Gunes, A., Inal, A., Taskin, M.B., Sahin, O., Kaya, E.C. & Atakol, A. (2014). Effect of phosphorus–enriched biochar and poultry manure on growth and mineral composition of lettuce (Lactuca sativa L. cv.) grown in alkaline soil. Soil Use and Managment. 30:182–188. doi: 10.1111/sum.12114
Hasana, H., Beyene, S., Kifilu, A. & Kidanu, S. (2022). Effect of phosphogypsum amendment on chemical properties of sodic soils at different incubation periods. Applied and Environmental Soil Science 9097994. doi: 10.1155/2022/9097994
Hsieh, E.J. & Waters, B.M. (2016). Alkaline stress and iron deficiency regulate iron uptake and riboflavin synthesis gene expression differently in root and leaf tissue: implications for iron deficiency chlorosis. Journal of Experimental Botany 67:5671–5685. doi: 10.1093/jxb/erw328
Hussain, S., Zhang, J., Zhong, C., Zhu, L., Cao, X., Yu, S., James, A. B., Hu, J. & Jin, Q. (2017). Effects of salt stress on rice growth, development characteristics, and the regulating ways: A review. Journal of Integrative Agriculture 16(11):2357–2374. doi: 10.1016/S2095–3119(16)61608–8
Hussien Ibrahim, M. E, Adam Ali, A. Y., Zhou, G., Ibrahim Elsiddig, A. M, Zhu, G., Ahmed Nimir, N. E, & Ahmad, I. 2020. Biochar application affects forage sorghum under salinity stress. Chilean journal of agricultural research ٨٠(٣): ٣١٧-٣٢٥. doi: 10.4067/S0718-58392020000300317
International Biochar Initiative (IBI). (April 2012). Standardized product definition and product testing guidelines for biochar that is used in soil. International Biochar Initiative. https://biochar–international.org/
IRAM/SAGyP 29571–3 Environmental quality – Soil quality. Determination of organic matter in soils. Part 3 – Determination of organic carbon.
Ke, H., Zhang, Q. & Liu, G. (2018). Effects of wood biochar addition on growth of cherry radish (Raphanus sativus L. var. radculus pers) IOP Conf. Ser.: Earth Environ Sci. 128:012182. doi:10.1088/1755–1315/128/1/012182
Lehmann, J. & Joseph, S. (2015). Biochar for environmental management: an introduction. In: Lehmann J, Joseph, S editors. Biochar for Environmental Management Science and Technology. London: Routledge; p. 1–12.
Liao, S., Bo, P., Li, H., Zhang, D. & Xing, B. (2014). Detecting free radicals in biochars and determining their ability to inhibit the germination and growth of corn, wheat and rice seedlings. Environmental Science & Technology 48:8581–8587. doi: 10.1021/es404250a
Lizarazu, M. y Vega, A. S. (2012). Guadua. En: Zuloaga FO, Rúgolo de Agrasar ZE, Anton AM, Eds. Flora Argentina. Plantas Vasculares de la República Argentina. Monocotyledoneae: Poaceae: Aristidoideae–Pharoideae. 3(1):59–63.
Londoño, X. (2009). Usos y potencialidades de los bambúes en Sur América. Botánica Brasileira: futuro e compromissos: 236–243. 6º Congreso Nacional de Botánica, Feira de Santana, Bahia, Brasil.
Madrigal, L. P. (2016). Cambio climático, ensalitramiento de suelos y producción agrícola en áreas de riego. Terra Latinoamericana 34(2):207–218.
Maraseni, T. N. (2010). Biochar: maximising the benefits. International Journal of Environmental Studies 67(3):319–327. doi:10.1080/00207231003612225
Martínez de Zorzi, V. (2019). Adición de biocarbón de Guadua chacoensis (Poaceae, Bambusoideae, Bambuseae) en suelos contaminados y su efecto sobre el crecimiento de Arabidopsis thaliana (Brassicaceae). Thesis presented in fulfillment of the requirements for the degree in Environmental Science in the college of Facultad de Agronomía, University of Buenos Aires, Argentina.
Mete, F. Z., Mia, S., Dijkstra, F. A., Abuyusuf, M. & Hoissan, A. S. M. I. (2015). Synergistic effects of biochar and NPK fertilizer on Soybean yield in an alkaline soil. Pedosphere 25(5):713–719.
Munns, R. & Tester, M. (2008). Mechanisms of salinity tolerance. Annual Review of Plant Biology. 59:651–681. doi: 10.1146/annurev.arplant.59.032607.092911
Muñoz, C., Góngora, S. & Zagal, E. (2016). Use of biochar as a soil amendment: a brief review. Chilean Journal of Agricultural & Animal Sciences 32(1):37–47.
Naeem, M. A., Khalid, M., Ahmad, Z., Naveed, M. (2016). Low pyrolysis temperature biochar improves growth and nutrient availability of maize on typic calciargid. Communications in Soil Science and Plant Analysis. 47(1):41–51. doi:10.1080/00103624.2015.1104340
Nath, H., Sarkar, B., Mitra, S. & Bhaladhare, S. (2022). Biochar from biomass: a review on biochar preparation its modification and impact on soil including soil microbiology. Geomicrobiology Journal. 39:373–388. doi: 10.1080/01490451.2022.2028942
Pinheiro, J., Bates, D. & R Core Team. (2023). nlme: Linear and Nonlinear Mixed Effects Models. R package version 3.1–163, https://CRAN.R–project.org/package=nlme.
Rahman, M. M., Akter, N., Karim, M. A. & Hossain, M. M. (2017). Effect of salinity on germination, seedling growth and yield of radish (Raphanus sativus L.). Journal of Agriculture and Ecology Research International 12(2):1–10.
R Development Core Team. (2019). R: a language and environment for statistical computing. Vienna, Austria.
Rogovska, N., Laird, D., Cruise, R., Trabue, S. L. & Heaton, E. A. (2012). Germination tests for assessing biochar quality. Journal of Environmental Quality 41(4):1014–1022. doi: 10.2134/jeq2011.0103
RStudio T. (2019). RStudio: Integrated Development for R
Salem, T. M., Refaie, K. M., Sherif, A. E., Ahmed, M. (2019). Biochar application in alkaline soil and its effect on soil and plant. Acta agriculturae Slovenica 114(1):85–96. doi: 10.14720/aas.2019.114.1.10
Sanchez, R. M., Dunel Guerra, L. y Scherger, M. (2015). Evaluación de las áreas bajo riego afectadas por salinidad y/o sodicidad en Argentina. Programa Nacional Agua https://www.repositorio.cenpat-conicet.gob.ar/handle/123456789/566
Sangiacomo, M., Garbi, M. y Del Pino, M. (2002a). Manual de Producción de Hortalizas. Cultivo de rabanito – Producción Vegetal III – Horticultura http://www.hort.unlu.edu.ar/sites/www.hort.unlu.edu.ar/files/site/Rabanito.pdf
Sangiacomo, M., Garbi, M. y Del Pino M. (2002b). Manual de Producción de Hortalizas. Cultivo de rúcula – Producción Vegetal III – Horticultura http://www.hort.unlu.edu.ar/sites/www.hort.unlu.edu.ar/files/site/R%C3%BAcula.pdf
Sathe, P. S., Adivarekar, R. V. & Pandit, A. B. (2021).Valorization of peanut shell biochar for soil amendment. Journal of Plant Nutrition 45:(4):503–521. doi: 10.1080/01904167.2021.1963771
Shetty, R. & Prakash, N. B. (2020). Effect of different biochars on acid soil and growth parameters of rice plants under aluminium toxicity. Scientific Research Publishing (SCIRP) 10:12249. doi: 10.1038/s41598–020–69262–x
Singh, B., Mei Dolk, M., Shen, Q. & Camps–Arbestain, M. (2017). Biochar pH, electrical conductivity, and liming potential. In: Singh B, Camps–Arbestain M, Lehmann J, editors. Biochar: A Guide to Analytical Methods. CSIRO, London; p. 23–38.
Sonmez, S., Buyuktas, D., Okturen, F. & Citak, S. (2018). Assessment of different soil to water ratios (1:1; 1:2,5; 1:5) in soil salinity studies. Geoderma 144(1–2):361–369. doi: 10.1016/j.geoderma.2007.12.005
Sumner, M. E. & Miller, W. P. (1996). Cation exchange capacity and exchange coefficients. In: Sparks, DL editor. Methods of soil analysis Part 3: Chemical methods. Soil Science Society of America Book Series 5. Madison, Wisconsin; p. 1201–1230.
Sun, Y., Gao, B., Yao, Y., Fang, J., Zhang, M., Zhou, Y., Chen, H. & Yang, L. (2014). Effects of feedstock type, production method, and pyrolysis temperature on biochar and hydrochar properties. Chemical Engineering Journal 240:574–578. doi: 10.1016/j.cej.2013.10.081.
Thiers, B. 2023. Index Herbariorum: A global directory of public herbaria and associated staff. http://sweetgum.nybg.org/ih/
Vega, A. S. & Cámara Hernández, J. (2008). La floración de Guadua chacoensis (Poaceae, Bambusoideae, Bambuseae) Revista Facultad de Agronomía UBA 28(2–3):107–110. Spanish.
Vega, A. S. & Rúgolo, Z. E. (2016). Guadua Kunth. En: Rúgolo, Z. E. editor. Bambúes leñosos nativos y exóticos de la Argentina. 1ª edición, Ed. Trama, CABA; p. 99–112.
Walkley, A. & Black, I. A. (1934). An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Science. 37(1):29–38.
Wang, Y., Wang, X., Liu, M., Wu, Z., Yang, L., Xia, S. & Zhao, J. (2012). Adsorption of Pb (II) from aqueous solution to Ni–doped bamboo charcoal. Journal of Industrial and Engineering Chemistry 19:353–359. doi: 10.1016/j.jiec.2012.08.024
Wang, Y., Xiao, X., Xu, Y. & Chen, B. (2019). Environmental effects of silicon within biochar (sichar) and carbon–silicon coupling mechanisms: a critical review. Environ Sci Technol. 53:13570–13582. doi: 10.1021/acs.est.9b03607
Weber, K. & Quicker, P. (2018). Properties of biochar. Fuel 217:240–261. doi: 10.1016/j.fuel.2017.12.054
Yang, W. & Zhang, L. (2022). Biochar and cow manure organic fertilizer amendments improve the quality of composted green waste as a growth medium for the ornamental plant Centaurea cyanus. Journal of Environmental Science and Pollution Research 29:45474–45486. doi: 10.1007/s11356–022–19144–8
Yu, H., Zou, W., Chen, J., Chen, H., Yu, Z., Huang, J., Tang, H., Wei, X. & Gao, B. (2019). Biochar amendment improves crop production in problem soils: A review. Journal of Environmental Management 232: 8–21. doi: 10.1016/j.jenvman.2018.10.117
Yu, J., Wang, Z., Meixner, F. X., Yang, F., Wu, H. & Chen, X. (2010). Biogeochemical characterizations and reclamation strategies of saline sodic soil in Northeastern China. Clean – Soil, Air, Water 38(11):1010–1016. doi: 10.1002/clen.201000276
Zhao, W., Zhou, Q., Tian, Z., Cui, Y., Liang, Y. & Wang, H. (2020). Apply biochar to ameliorate soda saline–alkali land, improve soil function and increase corn nutrient availability in the Songnen Plain. Science of the Total Environment 722:137428. doi: 10.1016/j.scitotenv.2020.137428
Zhang, K., Wang, Y., Mao, J., Chen, B. (2020). Effects of biochar nanoparticles on seed germination and seedling growth. Environmental Pollution 256:113409. Doi: 10.1016/j.envpol.2019.113409
Zhang, W. B. (2009). Process and properties of bamboo charcoal in utilization of bamboo. I. Bamboo technology training course for developing countries. China National Bamboo Research Center, Hangzhou, Popular Republic of China: 97–108.
Revista científica de la Asociación Argentina de la Ciencia del Suelo